
Detoxie for ICH M7
Classify pharmaceutical impurities with dual-system QSAR predictions, automated expert review, and quantitative read-across, all validated for regulatory submissions.
AI-driven mutagenicity assessment for confident ICH M7 decisions


FDA 21 CFR Part 11 and ANVISA RDC 658/2022 compliant from day one.
Fragmented tooling and data silos
Relying on disconnected systems for Expert and Statistical assessments leads to critical documentation gaps. Your team wastes valuable time reconciling conflicting outputs, managing format mismatches, and manually merging data, increasing the risk of transcription errors and regulatory pushback.
Complex data fragmentation
Regulatory rejection risk
Manual read-across searches, analogue selection, and narrative writing consume days per impurity.
This creates project-critical scheduling dependencies due to the scarcity of qualified toxicologists.
Experimental Ames data, carcinogenicity results, and structural alerts are scattered across public, proprietary, and literature sources.
Assembling a complete evidence package requires searching multiple databases before the assessment even begins.
FDA, EMA, and ANVISA (RDC 964/2025) demand OECD-compliant documentation and mechanistic justification.
Incomplete assessments or missing weight-of-evidence rationale trigger regulatory questions that delay approvals and require costly remediation.












The expert review bottleneck
ICH M7 demands rigorous assessment of every impurity above qualification thresholds.
Teams that rely on fragmented tools, manual reviews, or single-method predictions face delays, rework, and regulatory pushback.
The regulatory burden of mutagenic impurity control








Fragmented tooling and data silos
Relying on disconnected systems for Expert and Statistical assessments leads to critical documentation gaps. Your team wastes valuable time reconciling conflicting outputs, managing format mismatches, and manually merging data, increasing the risk of transcription errors and regulatory pushback.
Complex data fragmentation
Regulatory rejection risk
Manual read-across searches, analogue selection, and narrative writing consume days per impurity.
This creates project-critical scheduling dependencies due to the scarcity of qualified toxicologists.
Experimental Ames data, carcinogenicity results, and structural alerts are scattered across public, proprietary, and literature sources.
Assembling a complete evidence package requires searching multiple databases before the assessment even begins.
FDA, EMA, and ANVISA (RDC 964/2025) demand OECD-compliant documentation and mechanistic justification.
Incomplete assessments or missing weight-of-evidence rationale trigger regulatory questions that delay approvals and require costly remediation.












The expert review bottleneck
ICH M7 demands rigorous assessment of every impurity above qualification thresholds.
Teams that rely on fragmented tools, manual reviews, or single-method predictions face delays, rework, and regulatory pushback.
The regulatory burden of mutagenic impurity control









Expert System:
Knowledge-Based Structural Alert Assessment
Evaluates structures against 235 expert-defined alerts derived from established mechanistic toxicology and peer-reviewed literature.
Covers key DNA-reactive mechanisms, including electrophilic moieties, N-nitroso groups, and aflatoxin-like functionalities.
Incorporates both activating and deactivating alerts validated against 17,632 experimental outcomes with full PPV/NPV metrics.
Fully documented per OECD Principles 1-5 with ready-to-submit QMRF v.2.1 reports.
Predictive power: The Detoxie hibrid architecture
Achieve end-to-end mutagenicity and carcinogenicity assessment for pharmaceutical impurities with a dual-system architecture designed for mechanistic transparency and regulatory traceability.


In Silico Expert Review &
Integrated eMuDRA Read-Across


Statistical System: AI-Driven Mutagenicity Prediction

Utilizes a deep learning architecture (SMI-TED 289M) trained on 17,632 Ames test outcomes from OECD 471-compliant studies.
Identifies complex structure-activity relationships (SAR) beyond explicit alerts, capturing compounds missed by traditional rule-based systems.
Provides strain-specific predictions for all standard Ames strains (TA98, TA100, WP2 uvrA, etc.) with and without metabolic activation.
Explainable AI (XAI) & Feature Attribution Mapping
Computes per-atom and per-bond attributions to quantify the influence of individual moieties on the final mutagenicity prediction.
Generates visual maps where orange highlights indicate mutagenicity-enhancing regions and blue indicates attenuating features.
Delivers a transparent "waterfall" audit trail from baseline probability to the final prediction, ideal for regulatory reviewers.
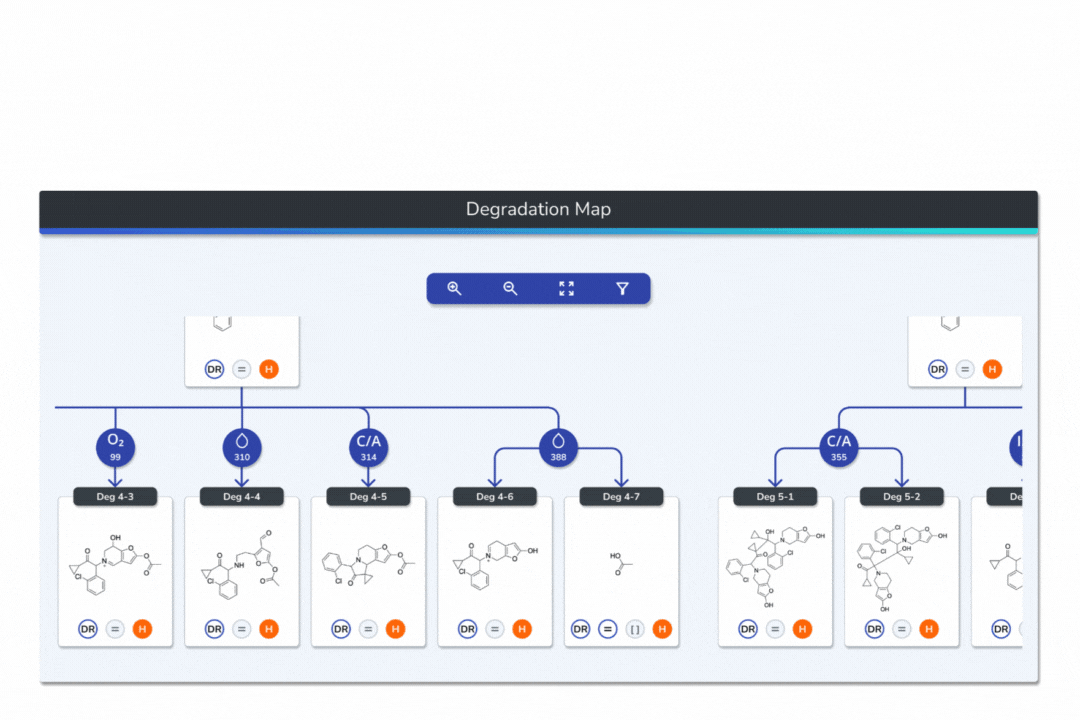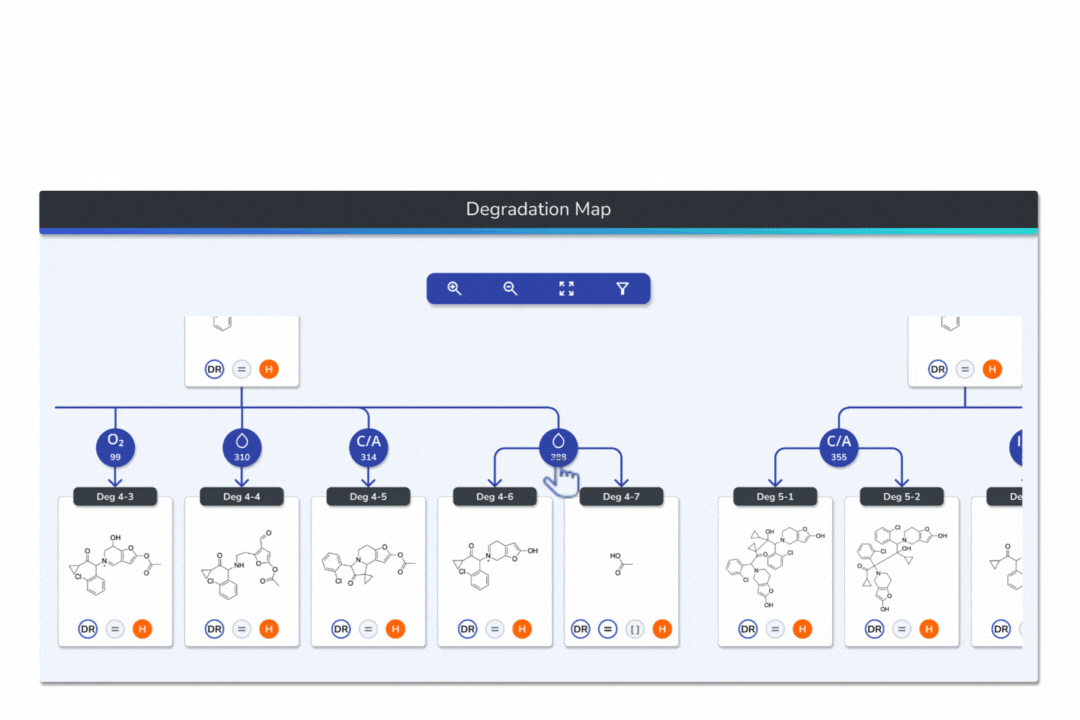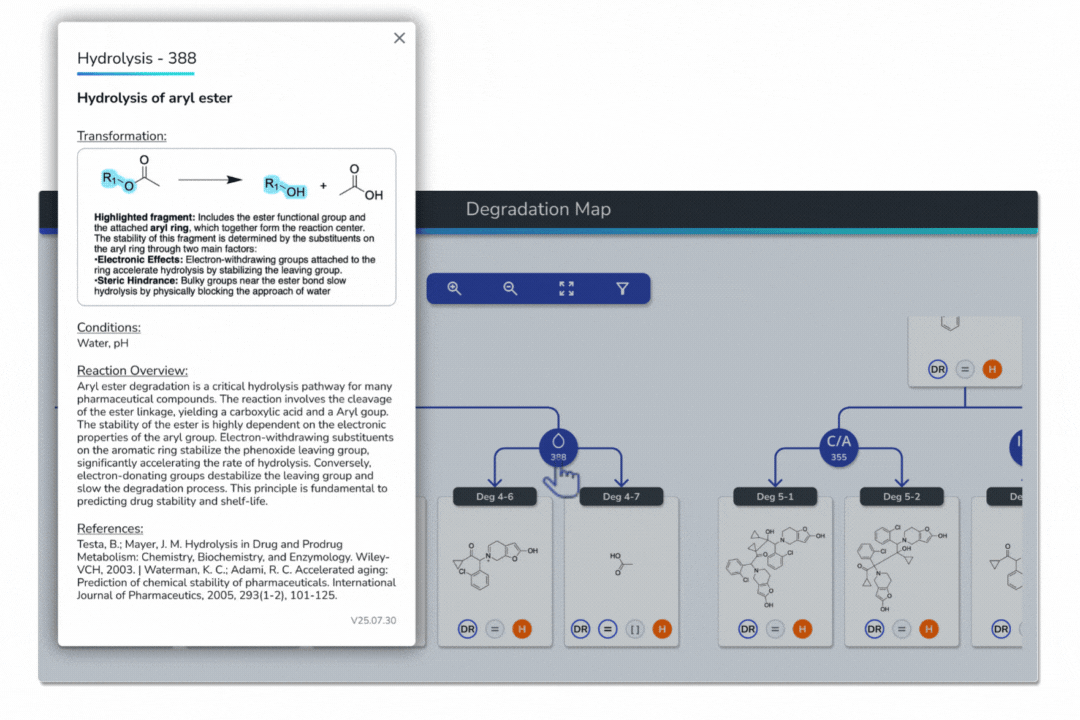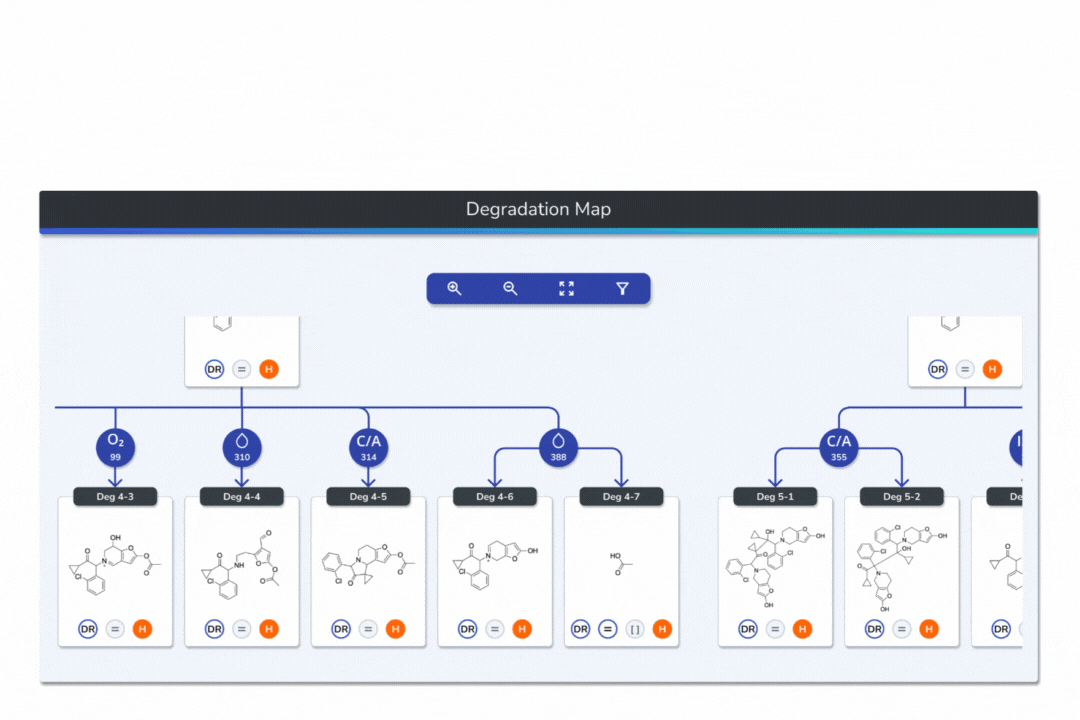

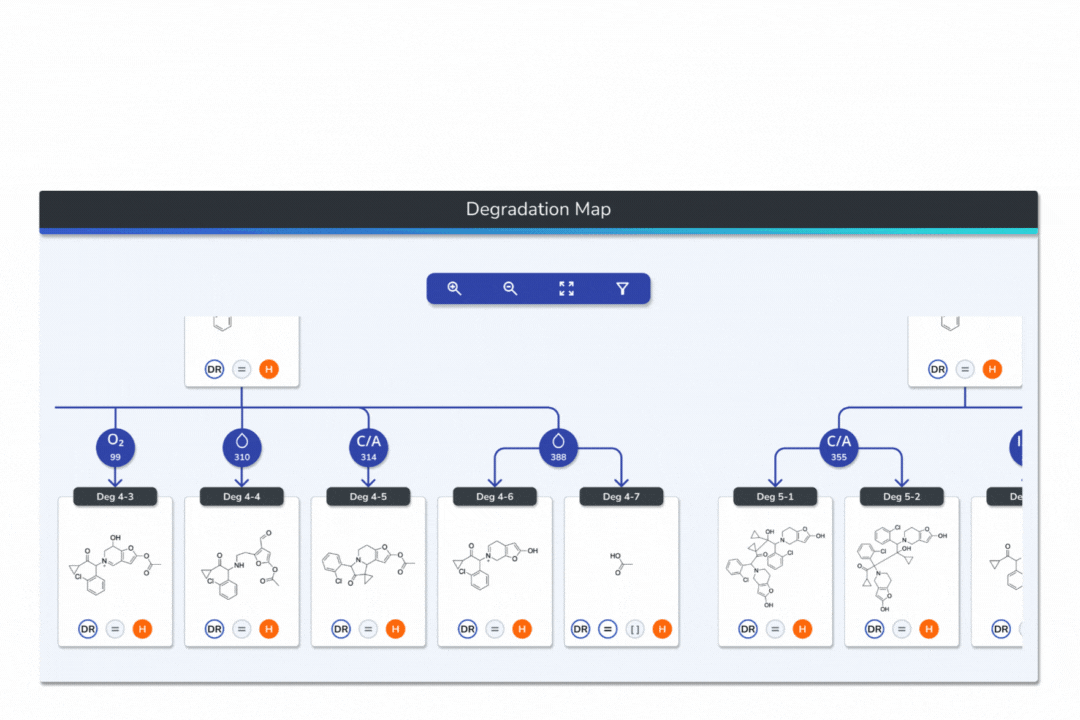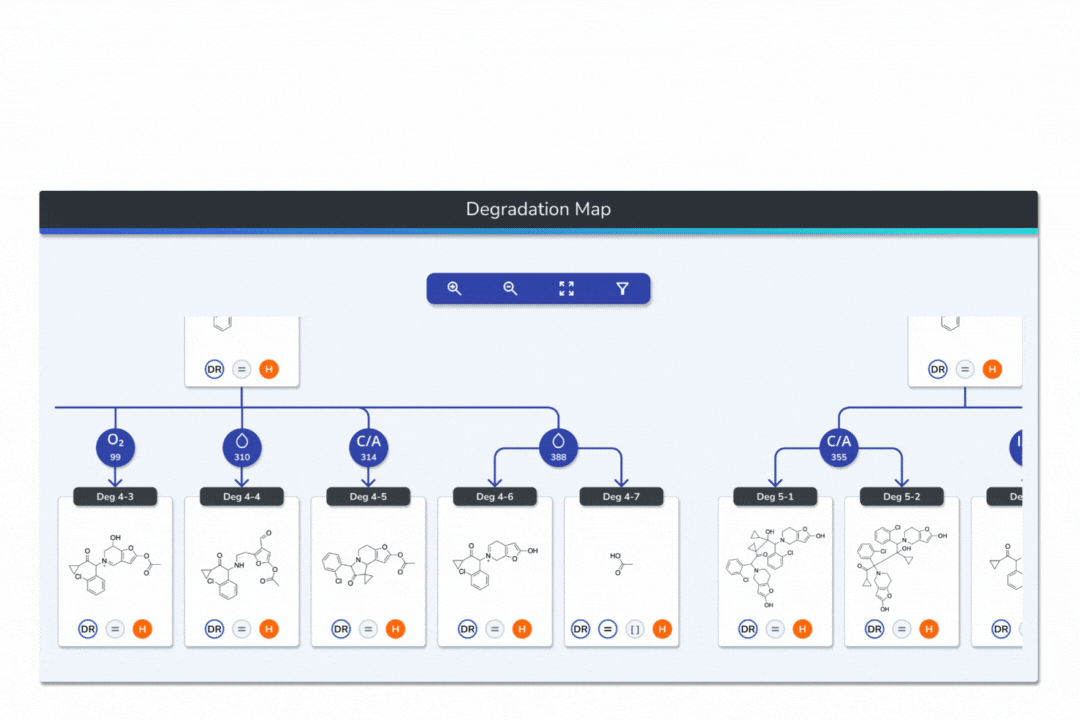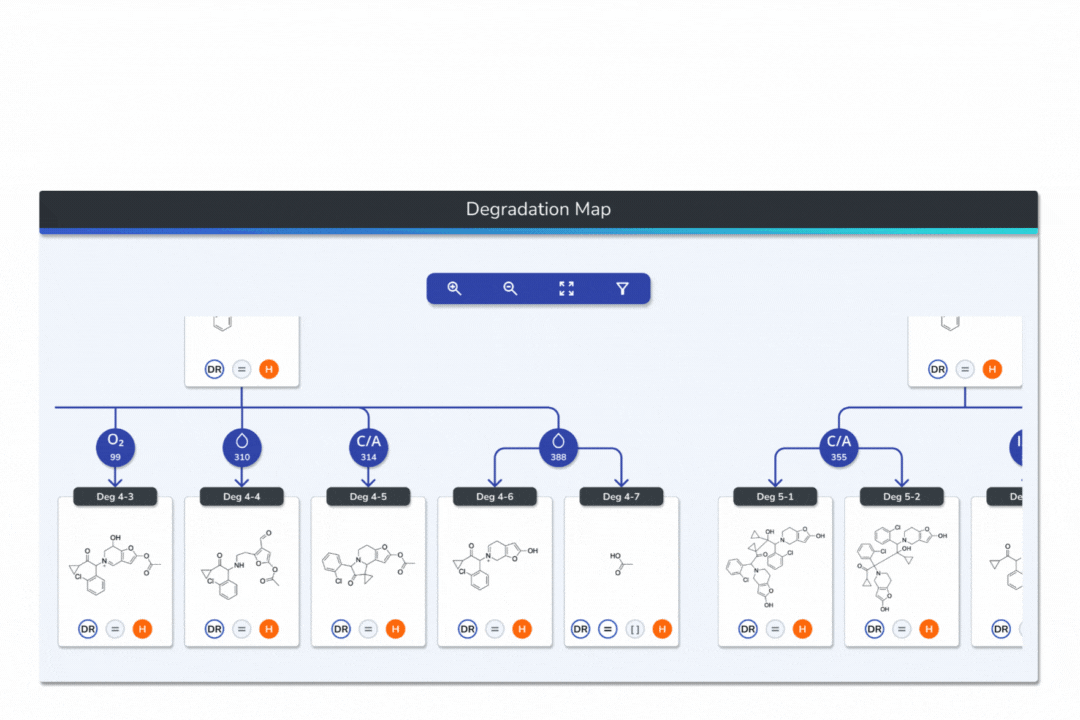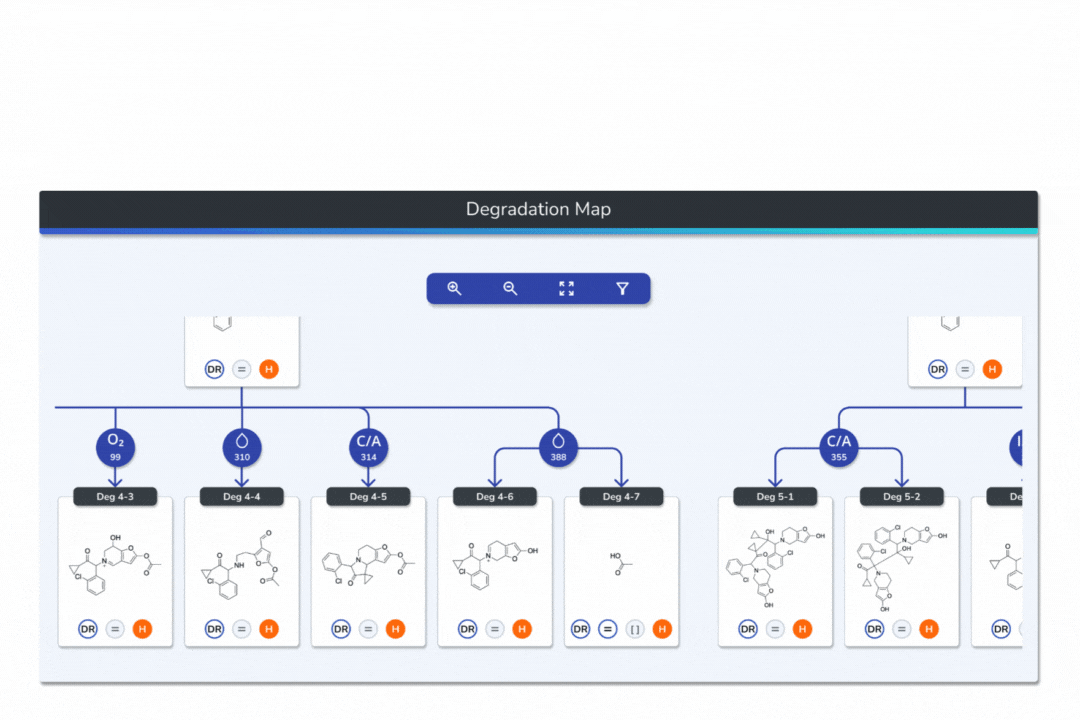
Features the built-in eMuDRA system for quantitative read-across to close structural analogues with verified experimental outcomes.
Evaluates analogue similarity through multiple descriptors following OECD Read-Across Assessment Framework (RAAF) principles.
Consolidates all evidence layers into a single weight-of-evidence table, assigning final ICH M7 Classes 1-5 and calculated AI values.
Provides full traceability from input structure to final classification, ensuring transparent communication with FDA, EMA, and ANVISA.
Automated Experimental Data & Carcinogenicity Retrieval
Instantly retrieves strain-specific Ames results from curated public databases, regulatory submissions, and peer-reviewed literature.
Integrates long-term rodent carcinogenicity data (OECD 451/453) including TD50 values and species-specific outcomes for 1,600+ compounds.
Ensures experimental data supersede in silico predictions per ICH M7 Section 5.2, while maintaining model outputs for cross-validation.
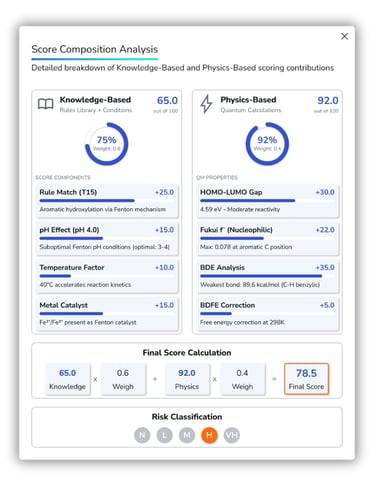


Regulatory-Grade Documentation
Generate immutable PDF reports with embedded QR codes.
Supports FDA 21 CFR Part 11, ANVISA RDC 658/2022, and GAMP 5 compliance requirements.
Comprehensive audit trails track all user actions and data modifications, ensuring data integrity and traceability for regulatory submissions.



Streamline your workflow by running both required (Q)SAR methodologies, Expert and Statistical, in a single platform. This integration removes the need for manual output merging, reconciliation between separate tools, or fixing format mismatches.
Both systems follow OECD principles and are ready for FDA, EMA, and ANVISA submission.
Why teams choose Detoxie
Unified ICH M7-compliant assessments
Accelerated expert decision support
Eliminate hours of manual searching with automated read-across from over 17,500 mutagenicity and 1,600 carcinogenicity results.
With XAI feature maps and pre-formatted Weight-of-Evidence summaries, toxicologists can review, affirm, and document conclusions significantly faster.
Replace blanket conservatism with structural alerts backed by performance metrics, mechanistic rationale, and the 12 most similar training compounds.
Explainable AI highlights specific molecular features driving each prediction, enabling targeted and precise impurity control strategies.
Transparent and mechanistic predictions




Regulatory-ready from day one
Ensure full compliance with built-in FDA 21 CFR Part 11, ANVISA RDC 964/2025, and GAMP 5 validation.
Immutable PDF reports with QR code verification and comprehensive audit trails integrate directly into quality management systems and regulatory dossiers.
Ensure gap-free regulatory submissions with transparent, mechanistic predictions and automated expert review.
Need more information? View Degrad Plus FAQ

Still have questions? Connect with our Experts.
If your specific scientific or regulatory questions remain unanswered, our team is ready to help.
Our Masters and Ph.D. experts can provide the detailed guidance you need or show you how Detoxie for ICH M7 empowers you to assess, classify, and justify every impurity with confidence.


